
One platform for MedTech. Total control.
MedTech clinical teams are lean by design. Most EDC platforms were built for large organizations with dedicated IT, data-managers, clinical operations and require months to implement.
The platform you choose should match the speed and agility of your team.
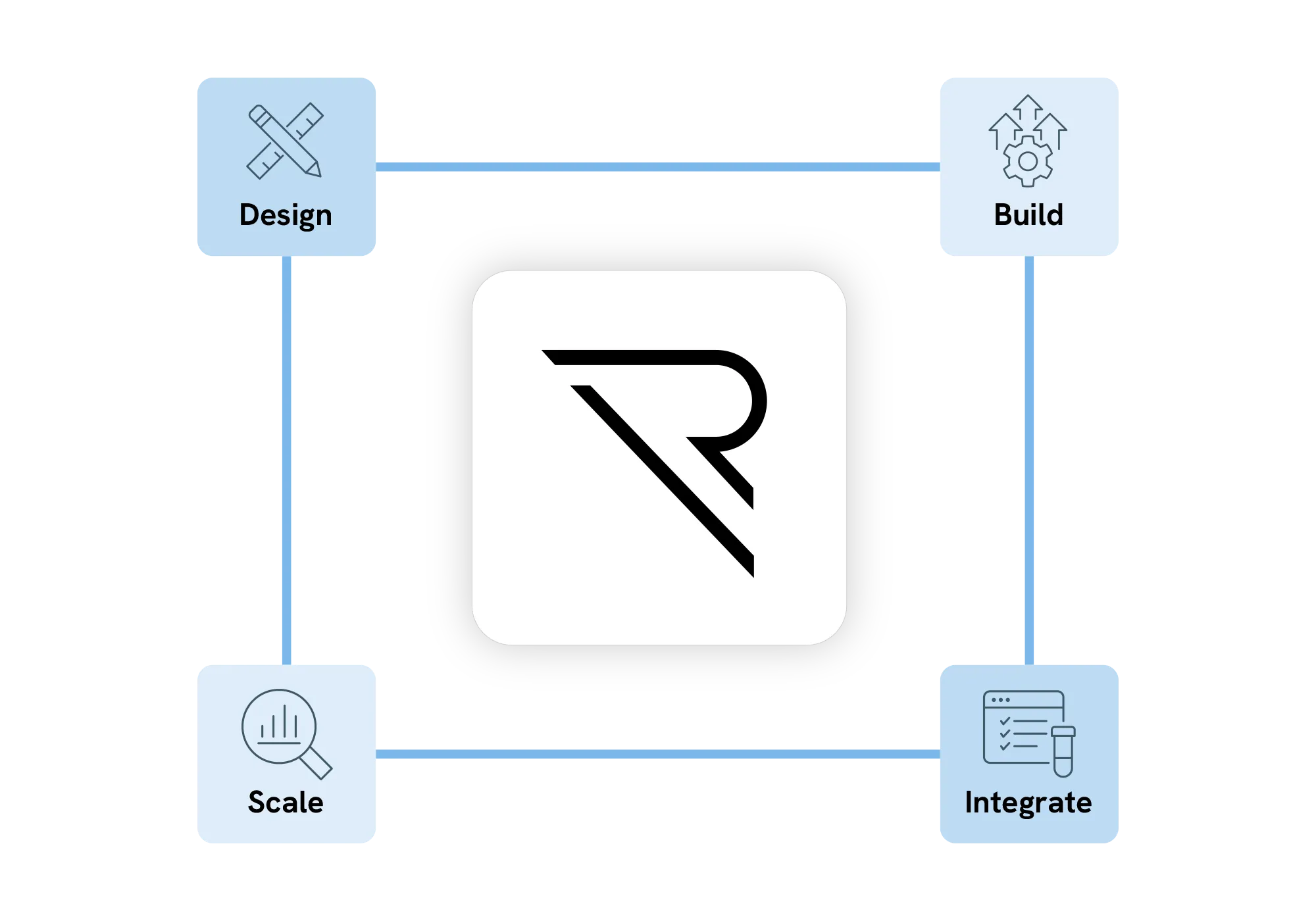
Build faster… effortlessly
REDCap Cloud gives medical device teams a single, validated platform to build, collect, and manage trial data. From first-in-human feasibility to post-market surveillance, all without the configuration complexity that kills timelines.
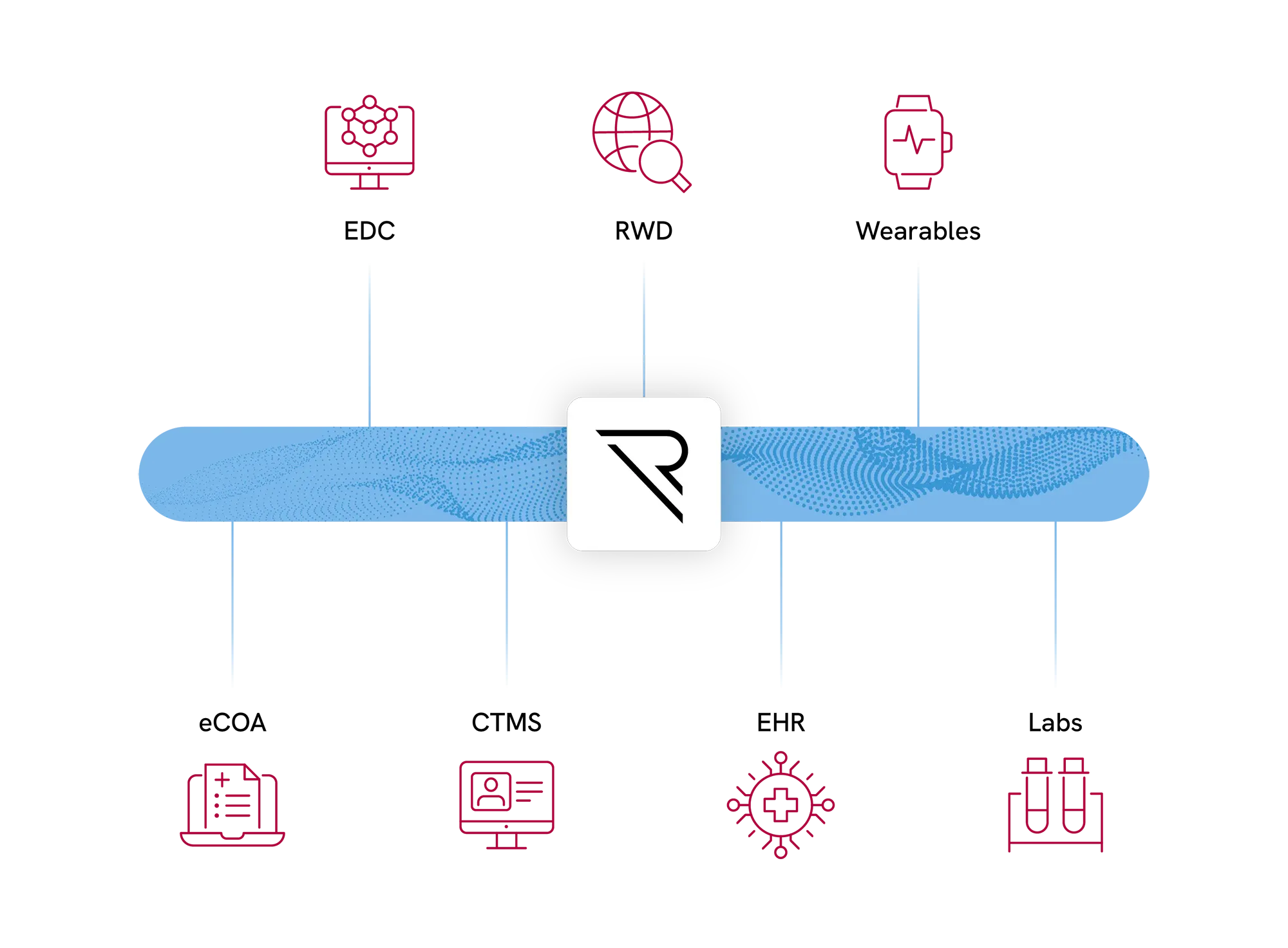
One unified platform. All your data.
Your device generates data from sources your EDC was never designed to handle. REDCap Cloud captures eCRF, ePRO, device readings, and lab feeds in one system — with bi-directional data flow that eliminates manual transfers, cuts SDV costs, and gives monitors real-time access to clean data from day one.
Compliance, technology and services to help across the MedTech lifecycle.
Pilot and Feasibility
Everything you need to get your first Pilot live.
- Start capturing data in days with REDCap Cloud’s self-service study builder — no IT team, no dedicated validator required.
- Add ePRO and eConsent modules as your study grows, without switching platforms or re-validating from scratch.
Pivotal
Full compliance and functionality.
- 21 CFR Part 11, ISO 14155, and MDR compliant. Audit trail active from first patient in, not retrofitted before submission.
- Configure device-native workflows with serial number tracking, malfunction logging, usability assessments, all without pharma terminology workarounds.
- Submission-ready CDISC exports, real-time monitoring dashboards, and Safety Gateway AE tracking all on one validated platform.
Post-Marketing Surveillance and PMCF
- Efficiently capture PMCF and post-market surveillance data through the same platform you used for your pivotal study without migration or revalidation.
- Run multiple PMCF studies simultaneously with flexible study templates and shared site networks.
- Real-world evidence collection built in with wearable integrations, patient diaries, and ePRO without adding vendors.







Book a Demo →
Start your journey with REDCap Cloud today – scale for tomorrows novel therapies